|
9/28/2023 0 Comments Western blot hiv example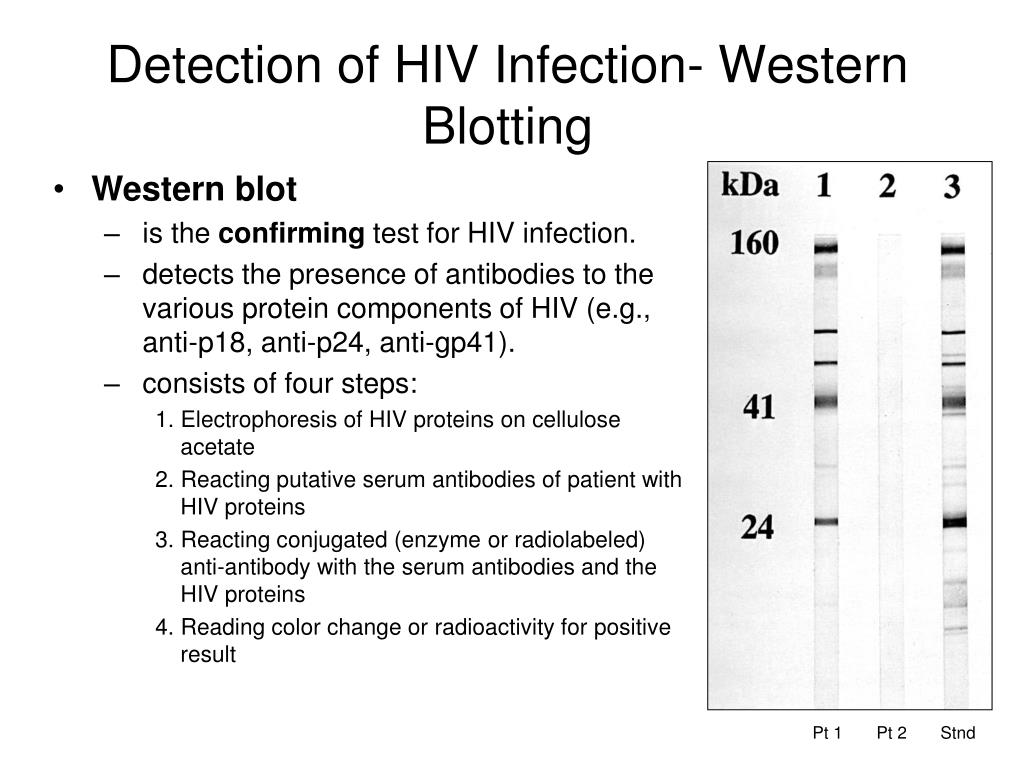
Jointly developed by CDC and The National Association of Community Health Centers, Inc. Implementation of Routine HIV Testing in Health Care Settings: Issues for Community Health Centers Practical guide to different approaches, considerations, and resources for making HIV testing routine in ED care. HIV Testing in Emergency Departments (Health Research and Education Trust) 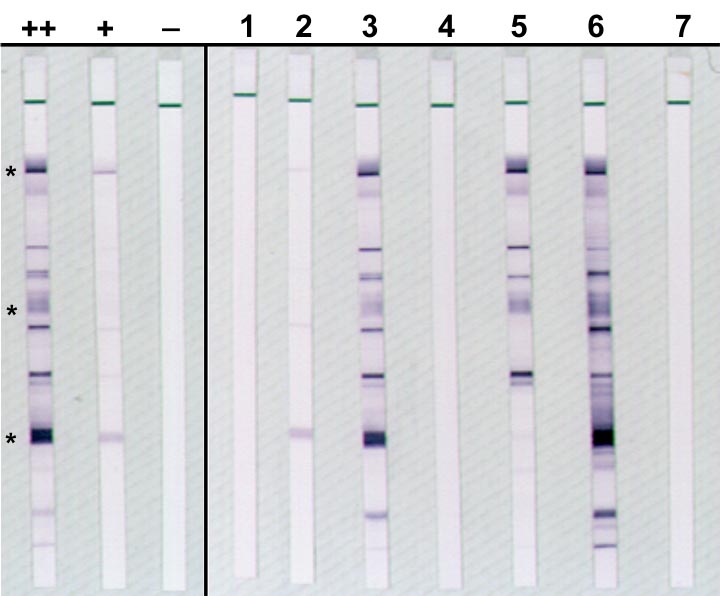
Provides information, exercises, and tools to design and implement a monitoring and evaluation (M&E) strategy for HIV testing and linkage programs in non-clinical settings. Serves as a companion resource to the Planning and Implementing HIV Testing and Linkage Programs in Non-Clinical Settings: A Guide for Program Managers. Note: Information on HIV testing may not be consistent with latest technologies.Įvaluation Guide for HIV Testing and Linkage Programs in Nonclinical Settings Supports planning, implementation, and evaluation of HIV testing and linkage services in non-clinical settings. Planning and Implementing HIV Testing and Linkage Programs in Nonclinical Settings: A Guide for Program Managers The purpose of the guide is to familiarize providers with key programmatic issues and updates that impact HIV testing service delivery in nonclinical settings.Ī CDC webpage that provides information and resources for providers and public on HIV testing in nonclinical settings. Grantee guidance to support the implementation of HIV testing services in nonclinical settings in the United States. Implementing HIV Testing in Nonclinical Settings: A Guide for HIV Testing Providers Implementation Resources Title and Description Technical Update on HIV-1/2 Differentiation Assays, August 2016.This technical update provides information on the use of the Alere Determine HIV 1/2 Ag/Ab Combo single use rapid test (Determine) in laboratories where it is not feasible to conduct an instrumented antigen/antibody test as the initial test in the algorithm. Technical Update Laboratory Algorithm for HIV Diagnosis, October 2017.*New Update* Technical Update for HIV Nucleic Acid Tests Approved for Diagnostic Purposes, May 2023.Provides updated recommendations for HIV testing by laboratories in the United States and describes approaches for reporting test results to persons ordering HIV tests and to public health authorities. Laboratory Testing for the Diagnosis of HIV Infection: Updated Recommendations Provides recommendations on routine, opt-out HIV screening of adults, adolescents, and pregnant women in health care settings in the United States. Revised Recommendations for HIV Testing of Adults, Adolescents, and Pregnant Women in Health Care Settings This MMWR policy note provides an update on CDC’s initiative to assess the benefits and harms associated with more frequent screening of MSM. Recommendations for HIV Screening of Gay, Bisexual, and Other Men Who Have Sex with Men - United States, 2017
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
